I answer an e-mail - Enhanced -
"Stars destroy work."
This was a short statement about finding the necessary complement to “work”, without which that “work” just would not be possible.
And, furthermore, without which nothing would exist, as “work” is physically a “parasitic” (thermodynamic or loss-generating), and not a “creative” (gravitational or gain-generating) process.
Note: The wind that bends the trees - or turns the windmills - is also performing "work".
You cannot use work to create something out of nothing (that isn't already there) - you can only convert it (within the laws of conservation of mass and energy).
On the other hand, gravity does this every second, everywhere - creating something out of nothing - and all it needs to do that is the matter, the property of which is itself.
Convoluted? Well, not really…
Now, DO stars ‘destroy work’ in turn?
Ultimately, yes, or rather its results, through their gravity.
Planets do so as well.
It just takes time.
Gravity decreases local and general entropy, the resulting work increases it, gravity decreases it again: 'Gravity works over time'.
Ultimately, nearly(?) all work is directed against gravity (which makes it possible in the first place by creating its conditions); nearly all the work we do - even just raising ourselves off the ground - is indeed directed against the force of gravity.
The same gravity that created everything, by the way.
Yes, this is where it approaches the religious... an almighty, omnipresent, eternal, omnipotent and independent force that creates what it will destroy and destroys what it has created, spawning fist-shaking dependents along the way, trying in vain to shake off their principal and eternal dependency.
The mind would boggle, if it weren't so simple.
Of course, these thoughts also revolve around the question of life and death, and the difference - and similarities! - between living and, such as a crystal or a machine, dead matter- which, through its gravity, provides not only for the historic primary source, the preconditions of appearance, but also the quasi- eternal, continuous source of upkeep for that living matter, i.e. the physical source of life, the eternal fountain of youth and renewal.
Other Voices
Physics of Life Reviews 5 (2008) 225–242
Charles H. Lineweaver, Chas A. Egan
Life, gravity and the second law of thermodynamics
There, for example, in the chapter ‘Physicists and life’, Ludwig Boltzmann is quoted with (life being) "a struggle for entropy” (emphasis and links added):
»Ludwig Boltzmann was concerned about entropy and the distinction between energy and free energy:
"The general struggle for existence of animate beings is therefore not a struggle for raw materials -these, for organisms, are air, water and soil all abundantly available- nor for energy which exists in plenty in any body in the form of heat (albeit unfortunately not transformable), but a struggle for entropy, which becomes available through the transition of energy from the hot sun to the cold earth."
[…However…]
In “What is life?” Schroedinger made it clear that Boltzmann’s animate beings were not struggling for entropy. If they were struggling at all, it was to get rid of entropy, or to absorb negentropy:
“What an organism feeds upon is negative entropy. Or, to put it less paradoxically, the essential thing in metabolism is that the organism succeeds in freeing itself from all the entropy it cannot help producing while alive.”
In the notes for a later edition Schroedinger apologizes to his physicist colleagues and admits that instead of negative entropy, he should have been talking about free energy.«
Leaving aside the concept of "free energy" mentioned there, which seems to be that particular bit of energy which is able to be "freed" from a potential under certain circumstances of inequilibrium - I don't believe energy comes with qualifiers such as "free" and “unfree” or "captive" (other than perhaps 'potential' and 'kinetic') - what we see here is the confusion which surrounds the concept of entropy:
One eminent scientist states that living creatures need entropy to be able to move (and thereby live), the other that these need negative entropy or negentropy (which, by the way, does not physically exist) to do so - as by the way so do machines; the same oxygen-starved carbohydrates (such as vegetable oil) plus free oxygen will run both - i.e. the exact opposite.
But it does not stop there
Taken and compiled from different sources:
Along comes Max Planck to declare: What we cannot calculate, we simply set:
»In a perfect crystal, every atom is in its clearly defined place. Theoretically, this can only be found at absolute zero temperature. Every movement is frozen. Since entropy is a measure of the disorder of a system, Max Planck postulated:
The entropy of all perfect crystalline substances at absolute zero is zero: S(0) = 0.«
So, at T = 0,
Entropy S = Q/T = Q/0 = 0
Really? And would this not be a forbidden division by zero? And thus enforce 'infinite entropy' near zero Kelvin?
Not quite: for conveniently, as we go on,
»It is impossible to reduce the temperature of a system to absolute zero of 0 K (Kelvin) by any process with a finite number of individual steps (lowest temperature reached so far = 2*10[-5]K).«
»In 1912, however, Max Planck suggested to arbitrarily assign (!) the value zero to the entropy at absolute zero. This made it possible to measure (!) entropies at other points if one chooses absolute zero as the reference state.« (!)
[This, by the way, implies that there IS such a thing as absolute zero]
»According to Planck, the entropy of ideally crystallized, pure solids is set to zero at absolute zero (!) (mixtures or frustrated crystals, on the other hand, retain a residual entropy)«
The measurement of entropy is indeed a problem.
And if the minimum is zero, does it then have a theoretical maximum?
If not - is there then no such thing as utter chaos?
With the current formula of
Entropy S = Q/T,
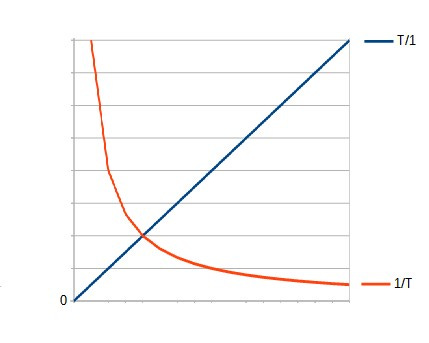
the envisaged division by zero, indeed, enforces the energy content Q to be zero as well at T=0 at least.With the "new" inverted formula of
Entropy S=T/Q,
at T=0 S=0 (independent of Q).
Physical Esoterics.
As in astrology vs. astronomy, that you are able to calculate within your system changes nothing:
A/B or B/A?
But that is what I suppose you end up with if you pick the “wrong” denominator, i.e. go for 1/x instead of x/1:
And, concerning the statement:
»It is impossible to reduce the temperature of a system to absolute zero of 0 K (=Kelvin) by any process with a finite number of individual steps.«
This, I suppose, would be the result of thermodynamic action (or “work”) with the resulting entropy always unequal to, or rather higher than zero, since "negative entropy" is impossible, as neither T (the absolute temperature) nor Q (the energy content) in S = Q/T can individually become negative in themselves.
But what about mass = 0, i.e. empty space? The result of a gravitational event? At what temperature is 'nothing'?
(Of course, empty space has no energy content either (or does it?); but this means that the entropy of 'nothing' -empty space- in that case is also 0.
Picking the issue up again
What do humans / living beings need and seek (within their boundaries)?
High Q/T or low Q/T (i.e. high T/Q)?
I suppose it would be high energy potential (potential free energy?) at low temperature:
High Q/T (or low T/Q)
(i.e., whole grain at room temperature; the other option, low Q/T, would be a dying fire of burnt grain - or, at a lower temperature, partly depleted, i.e. already digested grain, which could then be depleted fully by burning),
or:
With Entropy S=Q/T (current formula):
We need & seek HIGH entropy, or high energy content to temperature ratio - which is contradictory to high entropy signifying enhanced chaos, loss, thermodynamic outcome & lowered potential, but:
Boltzmann is right: We need HIGH Entropy - but that's not the one its inventor Rudolph Clausius (and we) talk about; in fact it's the opposite.With Entropy S=T/Q (the proposed formula):
We need & seek LOW entropy (or low temperature to energy content ratio - which is no contradiction to low entropy signifying order, gain, thermodynamic prerequisite & high potential, and
Schrödinger is right: We need LOW Entropy; but this is not the formula its inventor Clausius (and he) uses; in fact it's opposite.
So, perhaps, a lot of problems may disappear into thin air if you turn the formula for entropy on its head - what we are allowed to do.
Astro- and geophysicist Thomas Gold, in his 1999 book "The Deep Hot Biosphere: The Myth Of Fossil Fuels" (the underlying theory of which being that gas and oil are not dead and fossilized organic matter, but were generated directly by the Earth's gravity - which I don't subscribe to as yet, even though it would fit) is quoted with (hyperlinks added):
"The earth generates its own heat from compression, gravitational sorting (!) and radioactive decay deep within its core and mantle"
This would stay within the concept that gravity
a) sorts things out, while
b) generating energy, thus
c) generating "order" and thereby
d) reducing entropy.
Of course, that would turn everything upside down - but only the human interpretation of reality, not reality itself.
(Ayn Rand)
Elsewhere,
Roger Penrose is paraphrased as:
"Penrose is saying (…) the infant universe must have been highly ordered. The Second Law of thermodynamics leads him to this conclusion."
Yes, if the universe goes from orderly to disorderly on a merciless path - but how would then, mental gymnastics aside, complex living beings form themselves out of primitive and lifeless materials, atoms and molecules?
In a world pitilessly going from order to disorder, unrelentingly dissolving itself with no way of (re-) creating itself via the properties and the power of gravity, humans would indeed have to be lesser descendents of mythological gods and other higher beings, instead of being enhanced versions of visibly more primitive primates.
Addendum
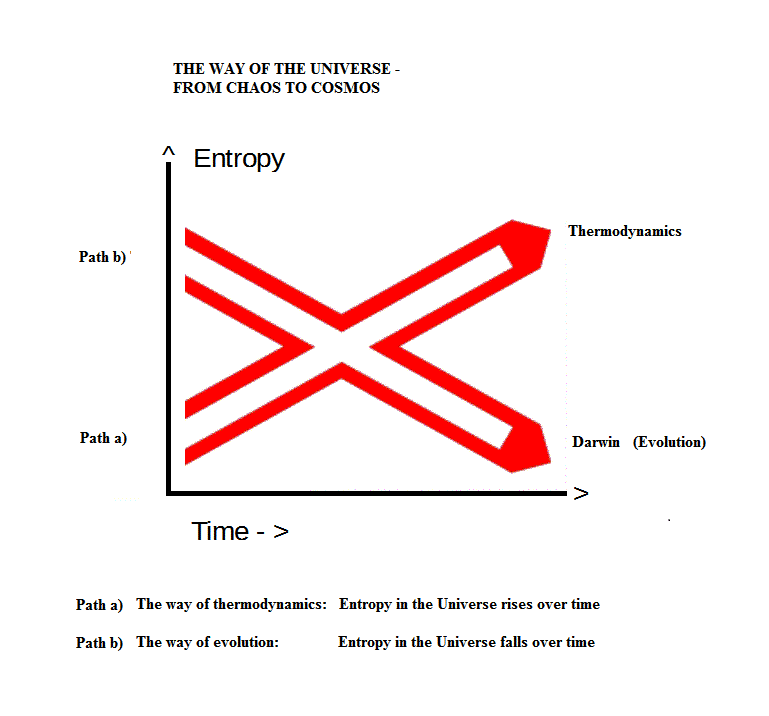
It seems there are two competing, if not contradictory, narratives concerning the development of entropy in the universe: one, that the measure of entropy rises over time, thus leading to infamous thermodynamic heat death of more or less absolute standstill; the other, that the evolution of living and even of dead matter constitutes a certain, if perhaps local, lowering over time of the measure of entropy.
Either which way you choose, you need a specific physical agent to provide for low entropy; either at the very beginning of time (path a) or over time (path b).
Barring God or gods as non-physical, there remains gravity.